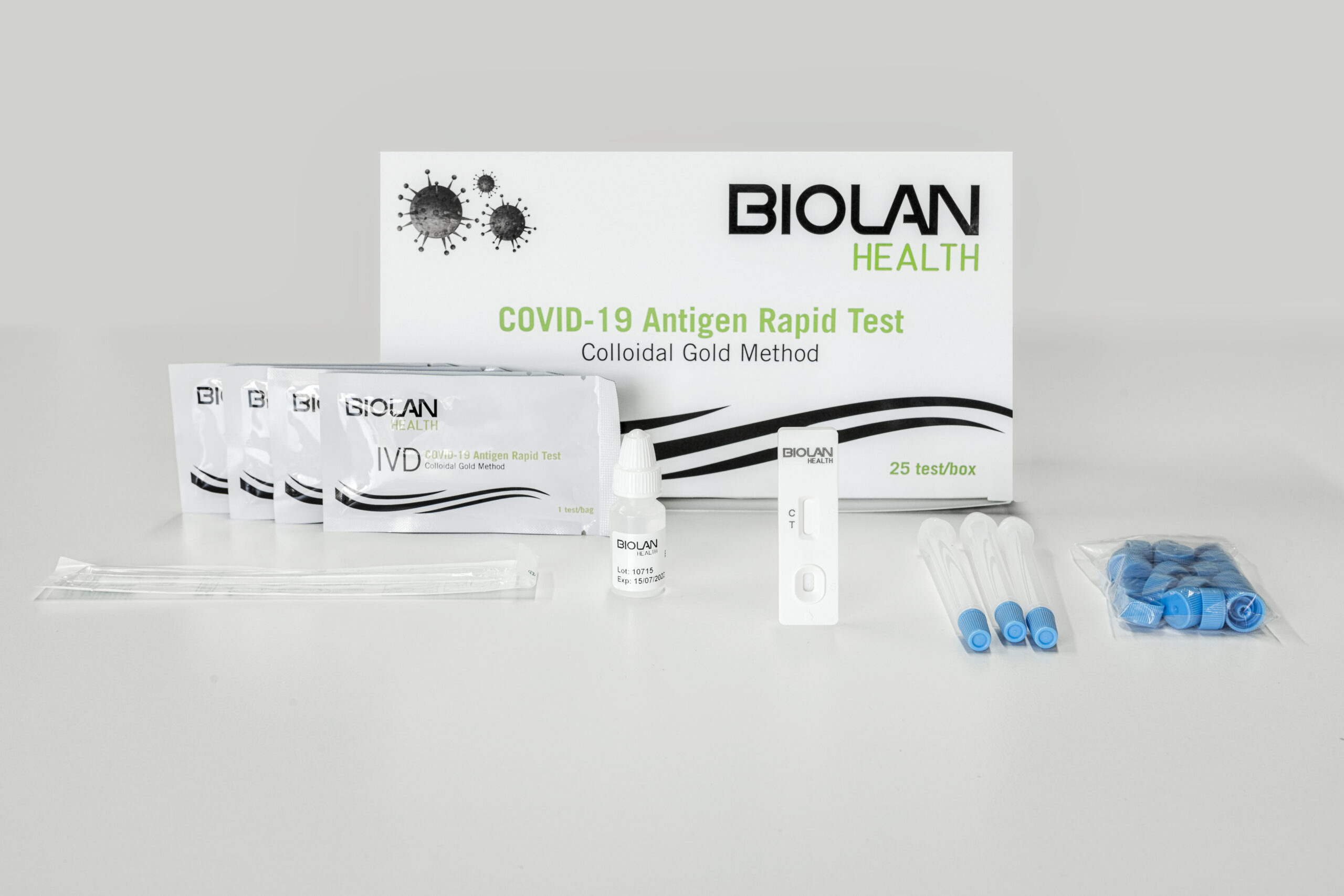
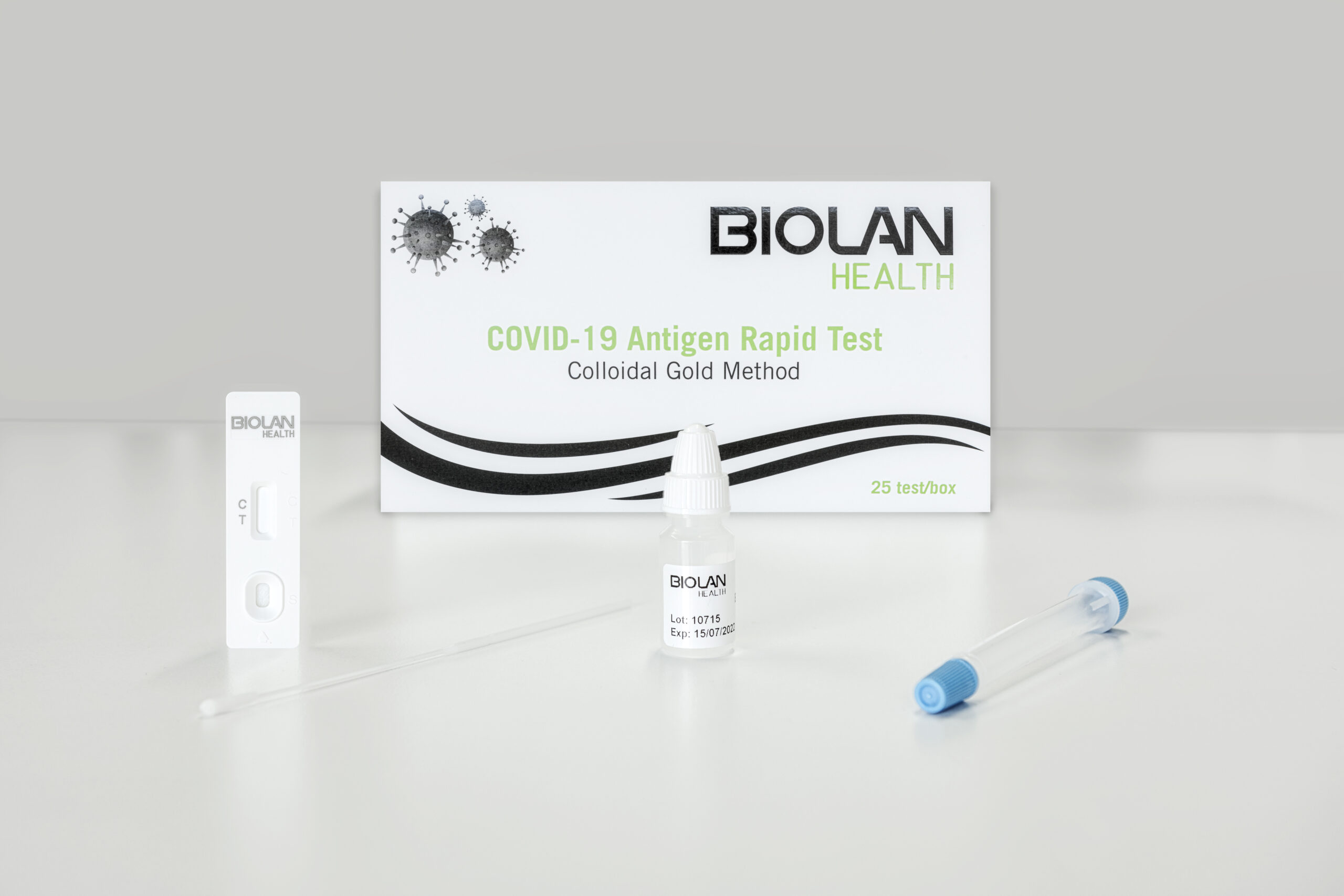
LATERAL FLOW IMMUNOASSAYBIOLAN HEALTH COVID-19 Antigen Rapid Test
- Easy sample collection with a nasal swab.
- Easy to use, no special equipment is required.
- Quick and accurate: results in 15-20 minutes.
- Sensitivity of 98.1%.
- Specificity 98.1%.
- Kit of 25 single-use tests.
Description
The BIOLAN HEALTH COVID-19 Antigen Rapid Test is a lateral flow immunochromatographic assay intended for the qualitative detection of the nucleoclapsid antigen of SARS-CoV-2 virus in nasal swab specimens in only 15-20 minutes.
Why use the BIOLAN HEALTH COVID-19 Antigen Rapid Test?
- For the early identification of people who are infected with the virus that causes COVID-19.
- It is very easy to use and it does not require laboratory equipments, allowing decentralized testing.
- It is less invasive than other diagnostic tests. It only requires a nasal sample much easier and more comfortable to obtain compared to the nasopharyngeal or oropharyngeal samples.
- It enables fast decision making because it provides a reliable results on site and in only 15-20 minutes
- The COVID-19 Antigen Rapid Test is developed and manufactured by BIOLAN HEALTH.
Download Flyer of BIOLAN HEALTH COVID-19 Antigen rapid Test
If you are interested in receiving more information about the Antigen rapid test, please send an email to: sales@biolanhealth.com, or by filling in the following form.
Online order form
Frequently Questions about the Antigen Rapid Test for COVID-19.
Coronaviruses are a large family of viruses that usually only affect animals. However, some have the ability to be transmitted from animals to humans. SARS-CoV-2 coronavirus is a new type of coronavirus that can infect humans and was first detected in December 2019 in Wuhan city, Hubei province, China.
Although the incubation period for COVID-19 ranges from 1 to 14 days, on average, it takes 5–6 days since someone is infected with the virus for symptoms to show
The most common symptoms are fever, cough and shortness of breath. In some cases, there are clinical forms with digestive symptoms (diarrhoea, abdominal pain), neurological symptoms (loss of taste, loss of smell) and even skin signs.
It is an immunochromatographic test for the qualitative detection of viral antigens, indicating SARS-CoV-2 infection.
It detects active infection by identifying the presence of virus proteins. The sample is processed at the site of collection providing results within about 15 minutes. A positive result means that the virus is present and there is probably an ongoing infection.
It should be performed within the first 7 days of infection, when viral load is at its peak.
The sample is taken using a swab that is inserted through the nostrils, making it less invasive and less uncomfortable than nasopharyngeal testing.
Time in which the results are known. The PCR requires sending the sample to a central lab to perform the analysis and therefore waiting a few hours to get the result However, the BIOLAN HEALTH Antigen Rapid Test provides results within about 15 minutes.
Sensitivity to symptoms. The PCR test is able to detect the presence of the virus even with very low viral loads. The antigen test requires a higher viral load and is therefore, recommended for symptomatic people or for contacts of someone who has tested positive for COVID-19 (within the first 7 days after the contact).
Reliability. Both techniques are very reliable but PCR is able to detect viral loads in a non-infectious state.
Easy sample collection for the antigen test because it only requires a nasal swab.
Cost the antigen test is considerably cheaper than the PRC.