The evolution of the pandemic caused by the virus that produces COVID-19 has been linked to the development of different diagnostic tests to detect the disease, such as the Rapid Tests for the detection of antigens. The first Rapid Tests that we knew of were all for the exclusive use of health professionals. But with the need to increase the diagnostic capacity of Public Health Systems and to be able to control the transmission of the virus, the first self-diagnostic antigen tests appeared in pharmacies, intended by the manufacturer to be used by the general population.
Unlike professional antigen tests, these tests allow the lay user to perform and interpret the test result themselves.
But, how can an antigen test ensure that an “inexperienced” person can perform and interpret the test correctly? How is its use validated?
The key is in the usability study. In the validation process of a self-diagnosis test such as the COVID-19 Antigen Rapid Test. Self Test developed by BIOLAN HEALTH S.L., the clinical validation includes a usability study to assess the viability of the self-test in a representative sample of the target population by means of two exercises that analyse all the steps involved in carrying out the test, from understanding the instructions for use to performing the test and interpreting the results:
– The execution of the self-test
– Interpretation of the test
Exercise on the execution of the self-test
The purpose of this exercise is twofold. On the one hand, to assess how easy it is for lay users to read and understand the instructions for use, and, on the other hand, to assess their ability to perform the test correctly and obtain a valid test result. For this purpose, all participants are provided with the instructions for use and the other educational tools included in the study test (such as a quick guide or a video), as well as all the components of the kit, and are asked to perform the self-test on their own, from sample collection to interpretation of the results. During the performance, each participant is supervised by a qualified health professional (such as a nurse), who can be called upon for assistance if any questions arise at any step of the test. At this point, the supervisor studies possible errors, doubts or suggestions that may arise during the execution of the test and assesses the user’s understanding -for example, by means of forms-. Similarly, the incidence of these doubts and errors is analysed in order to assess a possible revision of the instructions for use.
The results are compared with those obtained with a test carried out by the health professional and the percentage of positive and negative concordance between both results is calculated in order to know the percentage of positive and negative cases correctly detected by the lay user.
Test interpretation exercise
This exercise aims to verify that lay users are able to read and interpret test results correctly. For this purpose, participants are asked to read and interpret the results obtained in several tests which, as shown in Figure 1, cover a wide range of possible results (valid positive, valid mild positive, valid negative and invalid). We then compare the readings made by the lay user with those made by the health professional, answering two of the questions that many of us ask ourselves: do the lay user and the health professional see the same number of coloured lines, and do they give the same diagnostic test result?
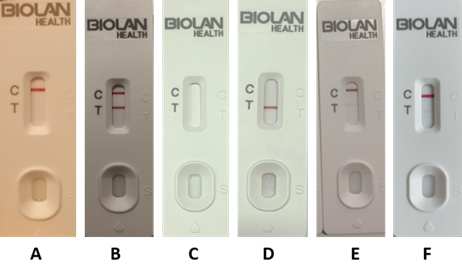
Figure 1: Example of tests used for the interpretation exercise. Negative test (A), positive test (B), invalid test (C and D) and mild positive test (E and F).
As in the previous exercise, participants can request help from the supervisor, who, in turn, assesses the difficulty of reading each of the tests and subsequent interpretation of the result in order to evaluate a new revision of the manual.
With the aim of offering the general population a new tool to help them detect a possible infection by the virus that causes COVID-19, BIOLAN HEALTH, in collaboration with HOSPITAL DE CRUCES, has carried out a usability study on 293 volunteers which has demonstrated the viability of the COVID-19 Antigen Rapid Test. Self Test of BIOLAN HEALTH, to be used as a self-diagnostic test by the general population.


